
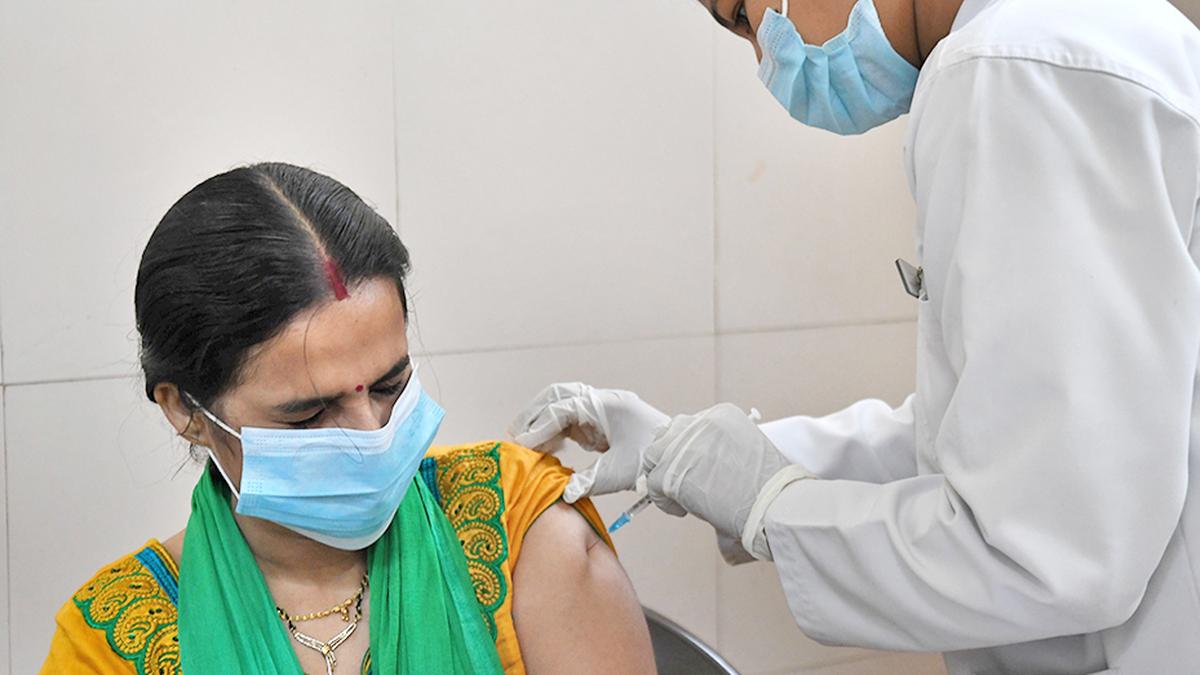
We now know why some people had severe blood clots after COVID-19 vaccines
In early 2021, as COVID-19 vaccines were being rolled out across the world, reports began to surface of a rare but alarming complication. Some people who received the shots were developing unusual blood clots. The cases were first identified in Europe and later in the U.S.
Notably, they were reported mainly among recipients of the AstraZeneca and Johnson & Johnson vaccines. The common link between those two vaccines was their design. Unlike the Pfizer and Moderna shots, which used mRNA, both the AstraZeneca and Johnson & Johnson vaccines used a modified virus to deliver DNA into the body’s cells. In roughly 3 to 10 cases per million vaccinated individuals, depending on age and sex, recipients developed unusual blood clots accompanied by low platelet counts, a condition that came to be known as vaccine-induced immune thrombocytopenia and thrombosis (VITT).
Very soon, research groups started reporting that the affected patients were producing antibodies against a human protein called platelet factor 4 (PF4). PF4 plays an important role in regulating the formation of blood clots. In these patients, antibodies were binding to PF4 and forming a complex that activated platelets, driving both clot formation and the low platelet counts.
However, the puzzling thing was PF4 is a human protein. The immune system is not supposed to make antibodies against self-proteins. In extremely rare cases, autoimmune reactions do occur due to genetic susceptibilities, but here, the vaccines were designed to generate immunity against the coronavirus spike protein, not against PF4. How could this be happening?
Delivering the recipe
At its core, a vaccine is essentially a decoy. It presents the immune system with something that looks like the enemy, so the system learns to recognise and defeat the real thing later. The goal of the COVID-19 vaccines was to teach the immune system to recognise the coronavirus’ spike protein. The vaccines do not contain the coronavirus itself. Instead, they deliver instructions that prompt our bodies’ own cells to briefly produce a harmless piece of the virus. The immune system sees this protein, mounts a response, and forms memory cells that stand ready for future encounters.
Cells store DNA inside a structure called the nucleus. When a protein needs to be made, the cell first creates a temporary working copy called messenger RNA (mRNA). The mRNA then exits into the main body of the cell, where special molecular machines called ribosomes make the protein from the mRNA. The mRNA is short-lived and quickly degraded.
mRNA vaccines such as Pfizer and Moderna took advantage of this system by delivering mRNA directly, packaged inside lipid (fat) particles. The mRNA never needed to enter the nucleus: it was immediately read in the cell body to produce the spike protein.
Delivering DNA is more complicated. DNA must enter the nucleus for the cell to make mRNA, which means crossing an additional protective barrier. Injecting naked DNA is inefficient.
Unique antibodies
Viruses, on the other hand, are experts at delivering DNA into cells. AstraZeneca and Johnson & Johnson used a harmless, genetically modified adenovirus as a courier to efficiently carry the coronavirus spike-encoding DNA into the nucleus, from where the cell’s own machinery took over.
Once the cell made the spike protein, it was displayed to the immune system, which began to respond. Among the first responders were B cells, the antibody-producing cells of the body. Each B cell carries on its surface a unique receptor, generated by a remarkable process of genetic reshuffling. During B-cell development, segments of DNA are randomly cut and pasted together in different combinations, creating millions of possible antibody designs. This process ensures enormous diversity even before an infection occurs.
When a B cell receptor recognises the spike protein, it is activated and begins to multiply. As it divides, its antibody genes undergo further fine-tuning through small mutations. Variants that bind the target more tightly are preferentially selected in a kind of microscopic evolutionary competition. Over days to weeks, this iterative cycle produces antibodies of increasing strength and specificity.
In theory, because this system relies on random recombination and mutation, every person’s antibodies are somewhat unique, even when facing the same virus.
Same single mutation
This is generally true for most antibodies. However, a new exception to this rule has been at the core of identifying the cause behind the anti-PF4 antibodies from patients with VITT. In a paper in the New England Journal of Medicine, investigators reported that antibodies isolated from patients from different countries — with no known connection to one another — were remarkably similar at the molecular level. These antibodies weren’t just targeting the same protein: they were built using the same antibody gene segments and carried highly similar structural features.
Even more intriguing: nearly all the affected patients shared one of two versions of an antibody gene, designated IGLV3-21*02 or *03. Additionally, in the process of fine-tuning, all the patients had generated the same single mutation, which led to a small change in the protein. This change, when coupled with the variations in two versions of the antibody genes, altered the electrical charge on that part of the antibody that binds to its target.
When researchers recreated these antibodies in the lab, they showed that this small alteration made a big difference. With the change, the antibodies stuck strongly to PF4 and activated platelets. When the change was reversed, the antibodies bound weakly and were far less likely to trigger clotting.
The researchers then turned to the next question: why this reaction occurred only with vaccines that used an adenovirus as the delivery vehicle. The answer to that lay inside the virus itself.

Cast a shadow
A protein within the adenovirus, called protein VII, contains a short stretch that closely resembles part of PF4. To the immune system, the entire adenovirus particle used for delivery was foreign and antibodies were naturally generated against its components. This is a known effect of these vaccines, and in almost all cases it is harmless. In mounting this response, the immune system first produced antibodies against protein VII. But as these antibodies were being refined, that critical change, in those individuals carrying one of the two specific antibody gene variants, altered their binding properties. As a result, the antibodies were mistaking PF4 for the viral protein and were reacting against the body’s own proteins instead.
For years, the mechanism behind VITT cast a shadow over adenoviral vector vaccines — a technology that has otherwise been central to global immunisation efforts. The new study has provided a clear molecular explanation by identifying protein VII to be the trigger and defining the precise antibody features involved. In so doing, the study’s authors — from Australia, Canada, Germany, and the Netherlands — have paved the way for future vaccines to be engineered even more carefully, further strengthening the safety of adenoviral vectors.
Arun Panchapakesan is an assistant professor at the Y.R. Gaitonde Centre for AIDS Research and Education, Chennai.
Published – March 10, 2026 07:15 am IST







