
How proteins are being tweaked to be quantum sensors inside the body
For decades, fluorescent proteins have been among the most powerful tools in biology. They glow when illuminated, allowing scientists to see where molecules are inside cells and how they move. From tracking cancer cells to mapping neural circuits, these luminous markers transformed the life sciences, work recognised with a Nobel Prize in 2008.
Now, two major studies published in Nature suggest that fluorescent proteins can do more than glow. Certain fluorescent proteins can be modified to detect magnetic fields and radio waves from inside living cells. In effect they behave as quantum sensors, devices whose operation depends on the behaviour of electrons at the smallest scales.
Until recently, quantum technologies were confined to ultra-cold laboratories filled with specialised equipment. Biology by contrast has been viewed as an unlikely home for quantum effects. Living cells are warm, crowded, and constantly in motion — conditions thought to destroy fragile quantum states.
The new results challenge that assumption, opening a path towards genetically encoded quantum sensors and a new class of hybrid quantum-biological technologies.
Hidden sensitivity
When a fluorescent protein absorbs light, one of its electrons is pushed into a higher-energy state. Usually, the electron quickly returns to its original state and releases energy as light. That simple process is what makes the protein glow.
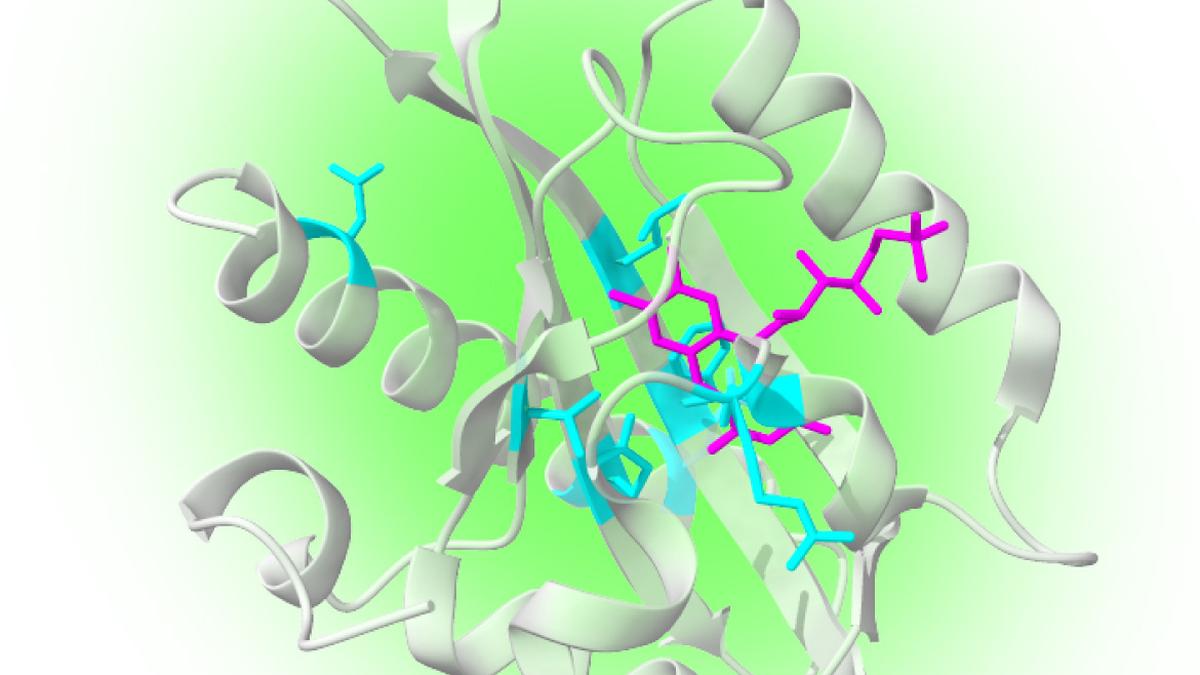
In some proteins, however, this journey is more complicated. The excited electron can briefly interact with a nearby molecule inside the protein, forming what scientists call a radical pair, two molecules that each carry an unpaired electron.
For a short time, the spins of these electrons are linked. The outcome of their interaction depends on weak magnetic influences around them. Even weak magnetic fields can change how the pair behaves, which in turn alters how much light the protein emits.
Chemists have known about this effect for decades, and it has been proposed as a possible explanation for how some animals sense Earth’s magnetic field. What had been missing was a way to reliably harness this phenomenon inside living cells.
Protein sensors
Researchers at the University of Chicago’s Pritzker School of Molecular Engineering focused on a variant of enhanced yellow fluorescent protein (EYFP), a close relative of green fluorescent protein. They discovered that EYFP possesses a metastable triplet state — a temporary electronic configuration in which an electron’s magnetic spin can be isolated and controlled.
Using carefully timed laser pulses, the team initialised the spin state of EYFP, manipulated it with microwave fields, and read it out optically, completing the full sequence required of a qubit.
They also detected optically driven magnetic resonance signals from EYFP inside living cells. These effects appeared in human kidney cells at low temperature and in Escherichia coli bacteria even at room temperature, showing that the protein’s quantum behaviour survives in biology’s noisy environment.
A second research group at the University of Oxford took a different approach. Working with a plant light-sensing protein, they used genetic engineering to create a family of magneto-sensitive fluorescent proteins called MagLOV. Through repeated rounds of mutation and selection, they produced versions with stronger and more stable magnetic responses.
The researchers showed that MagLOV proteins exhibit optically detected magnetic resonance in living bacterial cells at room temperature. In other words, radio waves at specific frequencies can predictably change fluorescence, directly revealing electron-spin behaviour.
Historically, biological candidates for quantum sensors were limited to purified, in-vitro systems, showed weak responses, or degraded quickly under light exposure. The engineered MagLOV proteins overcome many of these obstacles by combining stability, sensitivity, and genetic compatibility.
Together, the studies demonstrate that proteins can be programmed through DNA to act as quantum sensors.
Why inside cells matters
Most existing quantum sensors are made from solid materials such as diamond. These devices can be extraordinarily sensitive, but they are difficult to place inside cells or attach to specific biological targets.
Protein sensors are fundamentally different. Cells can produce them naturally once given the correct genetic instructions. The sensors can also be fused to other proteins, allowing researchers to position them at precise locations inside the cell.
This matters because many crucial biological processes involve subtle magnetic or electronic effects, including enzyme reactions with metal atoms, formation of short-lived free radicals, and electron transfer during respiration and photosynthesis.
Until now, studying these phenomena inside living cells has been nearly impossible. Protein-based quantum sensors offer a potential solution.
More than detection
The MagLOV researchers also showed that magnetic modulation can improve conventional fluorescence imaging. By switching a magnetic field on and off, they separated the MagLOV signal from background fluorescence and cellular autofluorescence. This technique, known as lock-in detection, enhances weak signals in noisy environments.
They further demonstrated a form of spatial localization based on magnetic resonance. Using magnetic-field gradients, they could determine the position of MagLOV-expressing cells within a three-dimensional sample, even when light scattering would normally blur the image.
The approach resembles some principles of magnetic resonance imaging (MRI), but uses genetically encoded fluorescent proteins as the signal source.

New ways of seeing
These studies point to a future in which genetically encodable quantum sensors reshape how scientists probe living systems. As sensitivities improve, protein-based qubits and magnetic-resonance probes could enable nanoscale measurements of magnetic fields, electric fields, temperature, and chemical environments directly inside cells.
Such sensors might track protein shape changes, monitor biochemical reactions in real time, or reveal how drugs bind to their targets with unprecedented precision.
Important challenges remain, though. Protein-based quantum sensors are currently less sensitive than solid-state devices, coherence times are shorter, and photobleaching remains a concern. Yet fluorescent proteins themselves took decades to become routine tools, and similar improvement could steadily close the gap.
Manjeera Gowravaram has a PhD in RNA biochemistry and works as a freelance science writer.
Published – February 23, 2026 05:30 am IST







